Feline Chronic Gingivostomatitis Educational Series: Part 1
What Causes FCGS? Understanding the Roots of a Painful Disease
If your cat has been diagnosed with feline chronic gingivostomatitis or if you’re a clinician watching a patient struggle through this painful, relapsing condition, one question keeps coming up: why? Why does the mouth become so inflamed that eating becomes agony? Why do some cats never fully recover even after all their teeth are removed? The answer involves a complex combination of viral infection, an altered oral microbiome, periodontitis, and a dysregulated immune system.
FCGS Is Not a Simple Infection
Feline chronic gingivostomatitis is a severely painful, debilitating oral inflammatory disease that affects cats worldwide. It is characterized by intense, diffuse inflammation of the gingiva and oral mucosa, extending far beyond what you’d expect from routine dental disease. Lesions can take an ulcerative or proliferative form, and many cats have both simultaneously.
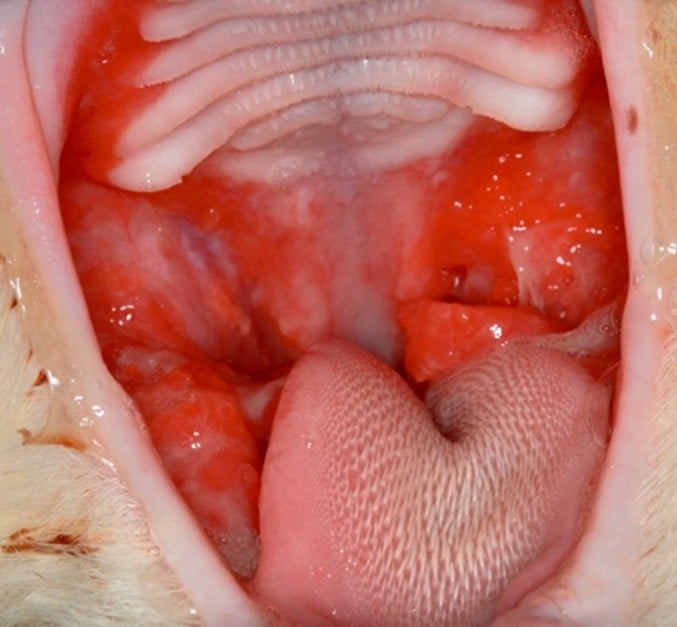
What makes FCGS particularly frustrating is that no single cause has been identified. Instead, current evidence points to a “perfect storm” scenario in which viral exposure, microbial imbalance, existing periodontal disease, and a dysregulated immune response converge, and the disease becomes self-perpetuating. Understanding each element helps explain why treatment is so challenging and why some cats respond to extraction while others do not.
FOR CAT PARENTS
FCGS is not caused by poor dental hygiene, and it is not your fault. It involves complex biological processes that researchers are still working to fully understand. What’s important is that early recognition and the right treatment approach will likely improve your cat’s quality of life.
Who Is Most Vulnerable?
FCGS tends to present in cats between 4 and 7 years of age, though it can occur at any age. While any cat can be affected, several environmental and infectious factors increase risk.
- Multi-Cat Households: Cats in shared households have 7 times greater odds of developing FCGS compared to single-cat homes. Each additional cat added to the household increases odds by more than 70%. Stress and pathogen transmission both likely contribute.
- Feline Calicivirus (FCV) Exposure: FCV is detected in 60–90% of cats with FCGS, making it the most consistently identified infectious risk factor. Shared spaces and social grooming facilitate transmission.
- Early-Onset Gingivitis: Young cats with persistent or worsening gingivitis at their first follow-up appointment appear to be at higher risk for progression to full FCGS, suggesting early dental disease may be a harbinger.
- Retroviral Infection: FeLV-positive cats are 7.5 times more likely to show no improvement after tooth extraction — a finding that underscores how underlying immune suppression can dramatically alter disease course.
The Mouth Before the Storm: Periodontitis as a Gateway
Periodontal disease and FCGS are intimately connected, but the relationship may be more directional than previously appreciated. Emerging data suggests that aggressive periodontitis can act as a precursor state, a transitional phase through which some cats pass before developing full-blown FCGS.
A 2025 case report using deep RNA metagenomic sequencing tracked a single cat’s oral microbiome as it progressed from aggressive periodontitis to FCGS. Researchers documented increasing microbial diversity, the emergence of periodontal pathogens like Porphyromonas and Treponema, and shifts in antimicrobial resistance and virulence genes, all consistent with a microbiome that had become progressively more hostile and less regulated.
This connection has practical implications: a young cat with aggressive gingivitis that isn’t resolving shouldn’t be dismissed as “normal dental disease.” It may be a window of opportunity to intervene before the immune system becomes entrenched in a chronic inflammatory cycle. Histopathological studies have confirmed that lesion severity correlates with the degree of immune cell infiltration in oral tissue — and in cats with FCGS, that infiltration can be profound.
CLINICAL PEARL
In cats presenting with early-onset gingivitis, lack of improvement at follow-up should raise the clinical suspicion for a disease trajectory toward FCGS. This may justify earlier, more aggressive monitoring and diagnostic workup.
An Ecosystem Out of Balance: Oral Dysbiosis in FCGS
The healthy feline oral cavity hosts a diverse but balanced community of bacteria and fungi. In cats with FCGS, that community is disrupted in ways that are both measurable and functionally significant — a state called oral dysbiosis.
Multiple studies have confirmed altered bacterial communities in the subgingival spaces of cats with FCGS compared to healthy cats and those with other oral diseases. Notably, the subgingival microbiome in FCGS cats appears to have prognostic value: cats with certain microbial profiles respond better to treatment than others. This suggests that which bacteria are present, not just how many, may affect outcome.
Fungi are also part of the story. The oral fungal microbiome (mycobiome) in cats with FCGS differs from that in healthy cats, pointing to a broader ecosystem-level disturbance rather than simply a bacterial problem.
What Dysbiosis Does to Tissue
Dysbiotic oral communities generate a more inflammatory local environment. Pathogens like Porphyromonas and Treponema, both associated with severe periodontal disease in multiple species, produce virulence factors that evade immune clearance and damage soft tissue. Studies using RNA metagenomic sequencing have found shifts in antimicrobial resistance as disease severity increases, suggesting that the microbial community is not just thriving in an inflamed environment — it may be actively contributing to and sustaining the inflammation.
WHAT THIS MEANS FOR TREATMENT
Full-mouth or partial-mouth tooth extraction reduces the microbial load significantly by removing the periodontal pockets that harbor pathogenic bacteria. This is why extraction remains a cornerstone of FCGS management. However, it does not fully “reset” the microbiome, which may help explain why 20–50% of cats remain refractory even after extraction.
The Viral Trigger: FCV and Beyond
Of all the factors associated with FCGS, feline calicivirus (FCV) carries the strongest and most consistent epidemiological signal. Between 60 and 90% of cats diagnosed with FCGS test positive for FCV, a striking prevalence that has long implicated the virus as a key driver of the disease.
Yet the relationship is not straightforward. FCV viral load does not correlate with the severity of oral lesions, nor does it predict treatment response to extraction. This tells us that the presence of FCV is not simply “causing” inflammation through direct cytopathic effect. Instead, it appears that the virus acts as a trigger or co-driver, provoking and perhaps sustaining an immune response that becomes dysregulated over time.
Feline Foamy Virus: A Complicating Factor
Feline foamy virus (FFV) is present in 20–80% of cats and generally does not cause clinical disease on its own. However, co-infection with both FCV and FFV is significantly more common in cats that are refractory to treatment, suggesting that this viral combination may adversely affect the immune system’s ability to resolve inflammation. FFV has also been shown to interfere with mesenchymal stromal cell (MSC) quality when used in regenerative treatment, a primary reason why allogeneic (healthy FFV free donor-derived) are preferred over autologous (patient-derived) MSCs.
FeLV and Immune Vulnerability
Feline leukemia virus infection represents a distinct but important viral risk factor. FeLV-positive cats show dramatically worse outcomes after dental extraction, likely because retroviral infection compromises the immune response needed to achieve and maintain remission. Cats with FeLV-associated FCGS also tend to have more lingual ulcers, another predictor of poor extraction response, creating a compounding disadvantage.
FOR VETERINARIANS
Retroviral testing (FeLV/FIV) and FCV PCR may be a worthy part of the diagnostic workup for any cat with suspected FCGS. Viral status directly informs prognosis after extraction and should be discussed transparently with owners when setting treatment expectations.
A Misfiring Immune System: The Central Pathology of FCGS
If the microbial and viral factors in FCGS are the triggers, immune dysregulation is the engine that keeps the disease burning. What distinguishes FCGS from ordinary gingivitis or periodontal disease is the nature and magnitude of the immune response in the oral mucosa — one that becomes self-sustaining and cannot be shut off through normal regulatory mechanisms.
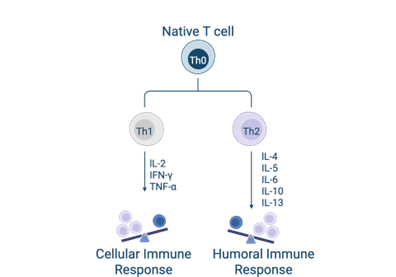
T Cell Imbalance
In healthy cats, oral mucosa is dominated by Th1-type helper T cells, which orchestrate controlled immune defense. In cats with FCGS, this shifts toward a mixed Th1 and Th2 cytokine expression profile, a transition that promotes chronic, tissue-damaging inflammation rather than efficient pathogen clearance. Adding to this, there is a significant reversal of the normal CD4/CD8 T cell ratio, with CD8+ cytotoxic T cells dominating the oral lesions. These cells are found in abundance in affected tissue and can contribute to direct tissue destruction.
Systemically, cats with FCGS show an increase in CD8+ effector memory T cells and a corresponding reduction in central memory T cells, a pattern that reflects ongoing, persistent immune activation rather than resolved infection.
The Cytokine Cascade
The inflammatory cytokine environment in FCGS is profoundly elevated. Tissue levels of IL-1β, IL-2, IL-4, IL-6, IL-10, IL-12, TNF-α, and IFN-γ are all increased. IL-6 appears to be a particularly influential driver: transcriptomic profiling of FCGS oral mucosa shows that gene expression patterns are heavily shaped by upregulated IL-6 signaling, with downstream effects on numerous inflammatory pathways.
There is also overexpression of IL-17A — the signature cytokine of Th17 cells. While Th17 cells play a protective role against microbial and viral pathogens, they are also implicated in inflammatory and autoimmune diseases. Their prominence in FCGS suggests a shift toward immune-mediated inflammatory pathways that may help explain why the disease persists long after the initial microbial or viral trigger has been reduced.
Antibody and Immunoglobulin Dysregulation
The humoral immune response is also markedly abnormal. Cats with FCGS have significantly elevated serum IgG, IgM, and IgA, consistent with chronic systemic antigenic stimulation, but paradoxically lower salivary IgA. Since salivary IgA plays a key role in mucosal immune defense and helps regulate the oral microbiome, its reduction may allow pathogenic organisms to persist unchecked at the mucosal surface, further sustaining dysbiosis and tissue damage.
Systemic Inflammation Beyond the Mouth
FCGS is not a purely local disease. Systemic markers of inflammation are consistently elevated, including peripheral neutrophilia and hyperglobulinemia. Alpha-1 acid glycoprotein, an acute-phase protein, is significantly elevated in FCGS cats and only partially decreases after dental extraction, suggesting that surgical intervention does not fully resolve the systemic inflammatory burden.
Notably, 98% of cats with FCGS also have concurrent esophagitis, a finding that underscores just how far the inflammatory process extends beyond the oral cavity. Vitamin D deficiency has also been documented in FCGS cats, which may further impair immune regulatory function given vitamin D’s known role in modulating immune responses.
Putting It All Together: A Disease of Converging Failures
What the current evidence describes is a disease that emerges from the convergence of several biological failures. A cat is exposed to FCV, potentially in a multi-cat environment, and the virus establishes chronic infection or recurrent reactivation in the oral mucosa. The resident microbial community shifts toward a more pathogenic composition, eroding the periodontal attachment and creating a local environment of sustained antigenic challenge. The immune system, already challenged by viral pressure, mounts an increasingly dysregulated response: cytokines amplify, T cell ratios invert, regulatory mechanisms falter, and what began as a protective inflammation becomes a self-perpetuating cycle of tissue destruction.
This framework explains several clinical puzzles. It explains why extraction resolves symptoms in most cats (removing the microbial reservoir breaks the antigenic stimulus) but also why 20–50% fail to recover – because their immune dysregulation has become autonomous and no longer requires the original trigger. It explains why viral status (particularly FeLV and co-infection with FFV) predicts outcome, because the immune system’s residual capacity matters enormously. And it explains why cats that don’t respond to extraction may benefit from immunomodulatory approaches that work downstream of the microbial trigger, directly addressing the misfiring immune response.
A NOTE FOR CAT PARENTS
If your cat has been diagnosed with FCGS, know that this is a recognized, well-studied disease, and that treatment options continue to advance. While full-mouth tooth extraction is often the most important first step, cats that remain refractory are not out of options. Speak with your veterinarian about the full picture: viral status, microbial testing, and whether advanced treatments such as MSC therapy might be appropriate for your cat.
References and Source Material
Key Reviews & Systematic Reviews
Sánchez-Vallejo M, Vélez-Velásquez P, Correa-Valencia NM. Feline chronic gingivostomatitis: a thorough systematic review of associated factors. Journal of Feline Medicine and Surgery. 2025;27(4):1098612X241310590.
Winer JN, Arzi B, Verstraete FJM. Therapeutic Management of Feline Chronic Gingivostomatitis: A Systematic Review of the Literature. Front Vet Sci. 2016;3.
Lee DB, Verstraete FJM, Arzi B. An Update on Feline Chronic Gingivostomatitis. Veterinary Clinics of North America: Small Animal Practice. 2020;50(5):973-982.
Soltero-Rivera M, Goldschmidt S, Arzi B. Feline chronic gingivostomatitis: current concepts in clinical management. Journal of Feline Medicine and Surgery. 2023;25(8):1098612X231186834.
Rivas IL, Soltero-Rivera M, Vapniarsky N, Arzi B. Stromal cell therapy in cats with feline chronic gingivostomatitis: current perspectives and future direction. Journal of Feline Medicine and Surgery. 2023;25(8):1098612X231185395.
Risk Factors & Epidemiology
Peralta S, Carney PC. Feline chronic gingivostomatitis is more prevalent in shared households and its risk correlates with the number of cohabiting cats. Journal of Feline Medicine and Surgery. 2019;21(12):1165-1171.
Kim DH, Kwak HH, Woo HM. Prevalence of feline chronic gingivostomatitis in feral cats and its risk factors. Journal of Feline Medicine and Surgery. 2023;25(1):1098612X221131453.
Silva M, Fernandes M, Fialho M, Mestrinho L. A Case Series Analysis of Dental Extractions’ Outcome in Cats with Chronic Gingivostomatitis Carrying Retroviral Disease. Animals. 2021;11(11):3306.
Sykes JE, Westropp JL, Kasten RW, Chomel BB. Association between Bartonella species infection and disease in pet cats as determined using serology and culture. Journal of Feline Medicine and Surgery. 2010;12(8):631-636.
Periodontitis & Early-Onset Gingivitis
Soltero-Rivera M, Vapniarsky N, Rivas IL, Arzi B. Clinical, radiographic and histopathologic features of early-onset gingivitis and periodontitis in cats (1997–2022). Journal of Feline Medicine and Surgery. 2023;25(1):1098612X221148577.
Shaw CA, Soltero-Rivera M, Profeta R, Weimer BC. Case Report: Shift from Aggressive Periodontitis to Feline Chronic Gingivostomatitis Is Linked to Increased Microbial Diversity. Pathogens. 2025;14(3):228.
Southerden P. Review of feline oral disease: 1. Periodontitis and chronic gingivostomatitis. In Practice. 2010;32(1):2-7.
Oral Microbiome & Dysbiosis
Anderson JG, Rojas CA, Scarsella E, et al. The Oral Microbiome across Oral Sites in Cats with Chronic Gingivostomatitis, Periodontal Disease, and Tooth Resorption Compared with Healthy Cats. Animals. 2023;13(22):3544.
Rodrigues MX, Bicalho RC, Fiani N, Lima SF, Peralta S. The subgingival microbial community of feline periodontitis and gingivostomatitis: characterization and comparison between diseased and healthy cats. Sci Rep. 2019;9(1):12340.
Krumbeck JA, Reiter AM, Pohl JC, et al. Characterization of Oral Microbiota in Cats: Novel Insights on the Potential Role of Fungi in Feline Chronic Gingivostomatitis. Pathogens. 2021;10(7):904.
Viral Infection
Thomas S, Lappin DF, Spears J, Bennett D, Nile C, Riggio MP. Prevalence of feline calicivirus in cats with odontoclastic resorptive lesions and chronic gingivostomatitis. Research in Veterinary Science. 2017;111:124-126.
Druet I, Hennet P. Relationship between Feline calicivirus Load, Oral Lesions, and Outcome in Feline Chronic Gingivostomatitis (Caudal Stomatitis): Retrospective Study in 104 Cats. Front Vet Sci. 2017;4:209.
Fried WA, Soltero-Rivera M, Ramesh A, et al. Use of unbiased metagenomic and transcriptomic analyses to investigate the association between feline calicivirus and feline chronic gingivostomatitis in domestic cats. AJVR. 2021;82(5):381-394.
Arzi B, Kol A, Murphy B, et al. Feline foamy virus adversely affects feline mesenchymal stem cell culture and expansion: implications for animal model development. Stem Cells Dev. 2015;24(7):814-823.
Immune Dysregulation & Biomarkers
Harley R, Helps CR, Harbour DA, Gruffydd-Jones TJ, Day MJ. Cytokine mRNA Expression in Lesions in Cats with Chronic Gingivostomatitis. Clin Diagn Lab Immunol. 1999;6(4):471-478.
Harley R, Gruffydd-Jones TJ, Day MJ. Salivary and serum immunoglobulin levels in cats with chronic gingivostomatitis. Veterinary Record. 2003;152(5):125-129.
Harley R, Gruffydd-Jones TJ, Day MJ. Immunohistochemical Characterization of Oral Mucosal Lesions in Cats with Chronic Gingivostomatitis. Journal of Comparative Pathology. 2011;144(4):239-250.
Vapniarsky N, Simpson DL, Arzi B, et al. Histological, Immunological, and Genetic Analysis of Feline Chronic Gingivostomatitis. Front Vet Sci. 2020;7:310.
Dolieslager SMJ, Lappin DF, Bennett D, Graham L, Johnston N, Riggio MP. The influence of oral bacteria on tissue levels of Toll-like receptor and cytokine mRNAs in feline chronic gingivostomatitis and oral health. Veterinary Immunology and Immunopathology. 2013;151(3-4):263-274.
Peralta S, Grenier JK, Webb SM, Miller AD, Miranda IC, Parker JSL. Transcriptomic signatures of feline chronic gingivostomatitis are influenced by upregulated IL6. Sci Rep. 2023;13(1):13437.
Soltero-Rivera M, Shaw C, Arzi B, Lommer M, Weimer B. Feline Chronic Gingivostomatitis Diagnosis and Treatment through Transcriptomic Insights. Pathogens. 2024;13(3):192.
Kouki MI, Papadimitriou SA, Psalla D, Kolokotronis A, Rallis TS. Chronic Gingivostomatitis with Esophagitis in Cats. Veterinary Internal Medicine. 2017;31(6):1673-1679.
Abbasi F, Azargoun R, Asri-Rezaei S. Evaluation of Serum Vitamin D and C Concentrations in Cats With Gingivostomatitis. Veterinary Medicine & Science. 2025;11(3):e70279.
Mestrinho LA, Rosa R, Ramalho P, et al. A pilot study to evaluate the serum Alpha-1 acid glycoprotein response in cats suffering from feline chronic gingivostomatitis. BMC Vet Res. 2020;16(1):390.


