Disease State
Prevalence
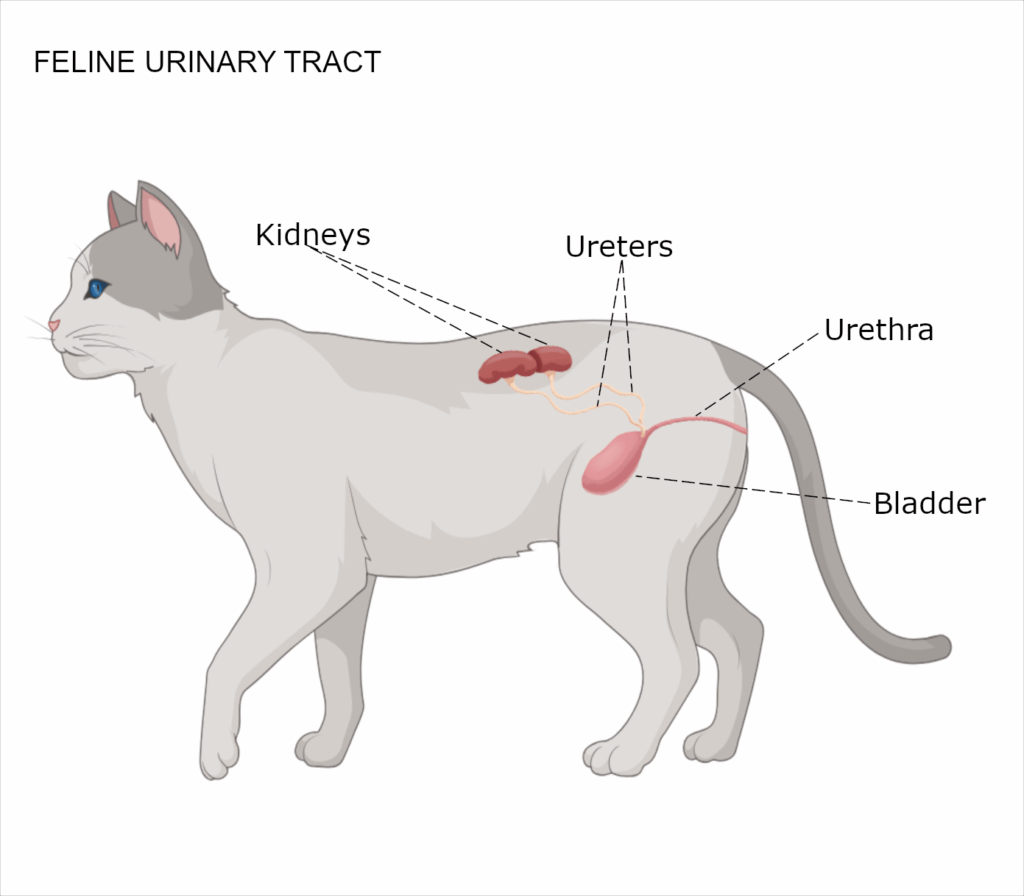
Chronic kidney disease (CKD) has an overall prevalence of up to 4% of cats. Prevalence of CKD increases with age, affecting approximately 40% of cats greater than 10 years of age and 80% of cats greater than 15 years of age.1,2 In cats greater than 5 years of age, CKD is the leading cause of death.1
Pathophysiology of CKD
CKD is defined as sustained (>3 months) reduction in glomerular filtration rate (GFR) or evidence of sustained (>3 months) kidney damage (i.e. structural damage, proteinuria). Feline CKD is histologically characterized by tubulointerstitial inflammation, tubular cell death, fibrosis, and reduction in blood vessel density.3 Oxidative stress has also been demonstrated in CKD cats.3 Complications including hypertension, anemia and proteinuria are common and often associated with disease progression and declining quality of life. Uremia associated with CKD has also been shown to have adverse effects on the gut microbiome in cats, however further studies are needed to understand the interplay between the fecal microbiome and serum concentrations of uremic toxins.4
Previous studies in humans have demonstrated that activation of inflammation is an important part of the pathogenic process of CKD, and that macrophage infiltration and aggregation contribute to the accelerated progression of CKD.5 CKD in cats has been shown to be associated with systemic inflammation, and new therapeutic approaches targeting inflammation appear warranted.6,7 Some cats with CKD also show changes in iron stores most consistent with functional iron deficiency, raising the question of whether iron supplementation is appropriate for cats with CKD.6
Clinical signs
The most common signs of feline CKD include polyuria/polydipsia (PU/PD), weight loss, and poor body condition. In more advanced stages, inappetence, lethargy, weakness, halitosis, and oral ulcers can occur. Systemic hypertension (defined as systolic blood pressure persistently >160–180 mmHg) affects approximately 20-40% cats seen in primary care practices8,9 and as many as 65% of cats in referral practice populations.10
Diagnosis
Diagnosis of CKD is typically performed using indirect methods which include measuring serum creatinine (sCr), blood urea nitrogen (BUN), urine specific gravity (USG), Symmetric Dimethylarginine (SDMA) and urine protein:creatinine (UPC) ratio. Anemia is also present in 30-65% of cats with CKD.11 Chronic anemia and the associated reduced oxygen perfusion to tissues negatively impacts the body in a number of ways, leading to an overall decrease in quality of life.11 Approximately 30% of cats with CKD have concurrent urinary tract infections (UTIs), however there does not appear to be an association between presence of a UTI and the severity of azotemia.12
Serum Creatinine (sCr)
Diagnosis is supported by documentation of a sustained sCr >1.6 over several weeks to months with clinical signs consistent with CKD.10 A persistent, substantial (>15%) increase in sCr from the cat’s historical baseline is also used as an indication of CKD. At least 67-75% of nephrons must be lost before increases in sCR levels are seen, therefore a change in sCR is considered a more sensitive marker in late course CKD than in early stage CKD.13,14 It should be noted that sCr has shown lower sensitivity as a marker of renal function in cats with hyperthyroidism.13
Blood urea nitrogen (BUN)
BUN concentration is increased in renal failure; however, in contrast to sCr, it is considered a less reliable indirect indicator of GFR due to its tubular re-absorption.14 BUN is also increased in several non-renal conditions including gastrointestinal bleeding and dehydration. Given its poor sensitivity and specificity as marker of renal function, it is generally recommended that BUN be evaluated in conjunction with sCr concentration and USG measurements.14
Urine Specific Gravity (USG)
CKD diagnosis is supported by documentation of a sustained USG < 1.035 over several weeks to months with clinical signs consistent with CKD.10
Symmetric Dimethylarginine (SDMA)
SDMA is a methylated form of the amino acid arginine. It is produced during protein turnover and is excreted almost exclusively by the kidneys, making it a good biomarker for kidney function, particularly GFR. SDMA has greater sensitivity than creatinine for detection of early CKD and does not appear to be affected by loss of muscle mass.10
Proteinuria
Proteinuria is associated with declining body condition and is considered to be a biochemical marker of kidney dysfunction.15
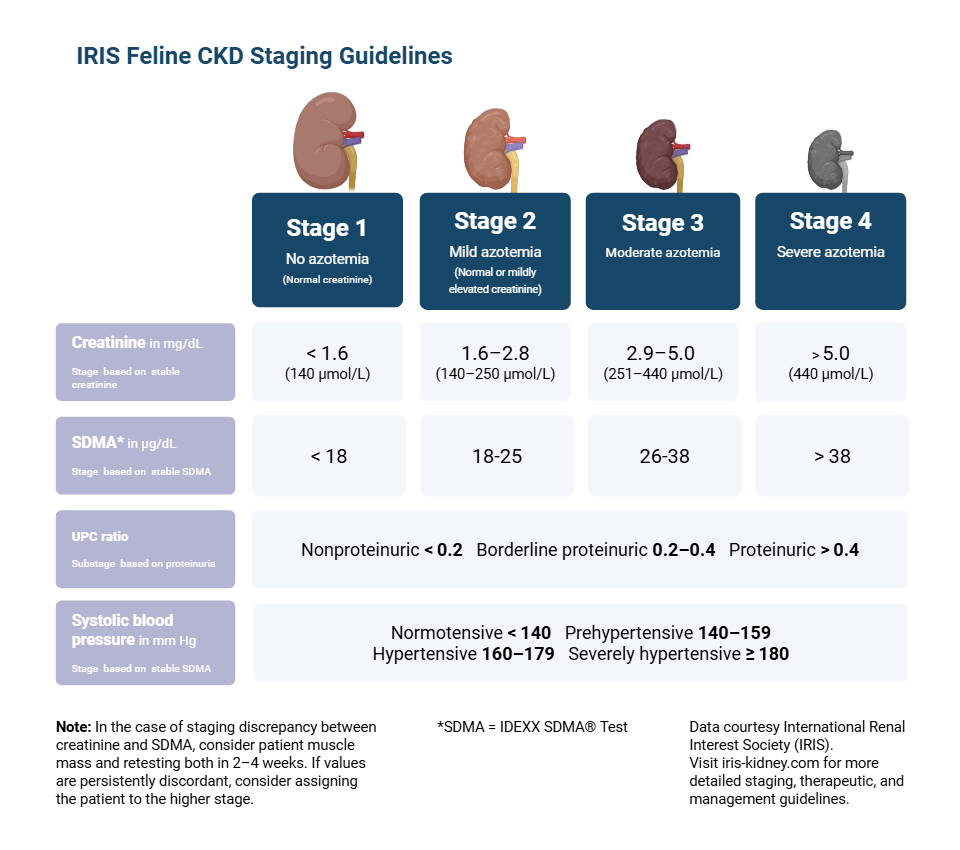
IRIS Staging
The International Renal Interest Society (IRIS) scoring system defines 4 stages of CKD based on sCr and SDMA measurements. UPC and systolic blood pressure measurements define substages of disease.
Glomerular filtration rate (GFR)
GFR is defined as the volume of ultrafiltrate produced by glomerular filtration per unit of time and can be measured using specific markers of urinary or plasma clearance.14 GFR is generally related to body mass and typically expressed in mL/kg/min. GFR is the most sensitive test to directly measure kidney filtration and excretion and detects the earliest decreases in kidney function. The most commonly used marker of GFR in human and veterinary medicine is iohexol, however direct GFR measurement methods are not frequently employed in routine clinical practice.16
Mechanism of Action
Current medical therapies
A multimodal approach is required to manage CKD and typically involves a combination of fluids to maintain hydration (encouraging drinking, subcutaneous fluid therapy), nutritional support through the diet (moderate protein, restricted phosphate and potassium supplementation), anti-hypertensives, anti-proteinurics, phosphate binders, anti-emetics and antacids. Other medications commonly used in CKD patients include Beraprost (prostacyclin analogue to slow kidney decline), Molidustat to treat anemia, and antibiotics to treat concurrent UTIs.
The burden of chronic medication administration in cats with CKD can be daunting, leading to alterations in the caregiver/cat bond and inconsistent dosing. In a UK study 2/3 of owners felt their lives changed a lot since their cat was diagnosed with CKD, 99% felt anxiety and other negative emotions about caring for their cat with CKD, and 37% felt caring for their cat negatively impacted or restricted their own life.17
Investigational Use of Mesenchymal Stem Cell Therapy in Feline CKD
Due to the limited treatment options for CKD beyond supportive care or transplantation, mesenchymal stromal (stem) cell (MSC) therapy is an emerging therapeutic approach being investigated for the treatment of CKD in humans and cats. MSCs have been shown to have anti-inflammatory, anti-fibrotic, and regenerative effects in CKD rodent models.18,19 MSC therapy offers the potential to reset kidney function by modulating the underlying pathophysiology and enhancing endogenous repair mechanisms.
Paracrine and Immunomodulatory Effects
MSCs exert profound immunomodulatory and anti-inflammatory effects via paracrine signaling, potentially playing a central role in slowing CKD progression. MSCs could modulate a wide array of pathological processes, including immune responses, apoptosis, oxidative stress, tubular epithelial–mesenchymal transition (EMT), and angiogenesis. 5 MSCs have been shown to secrete anti-fibrotic factors and regulate pro-inflammatory cytokines such as IL-6 and TGF-β, helping to reduce collagen deposition and suppress fibrosis. 5,20 MSCs also influence local immune cells, including macrophages, which promote anti-inflammatory effects in response to ischemic and hypoxic damage, tissue repair, and debris clearance. 20 In CKD rodent models, MSC therapy has resulted in improvement of several clinical biomarkers for CKD including body weight, creatinine, BUN, proteinuria, blood pressure, and hematocrit.3
Regenerative and Repair Capacities
Beyond immunomodulation, MSCs may directly contribute to renal tissue regeneration through several mechanisms. MSCs can differentiate into tubular epithelial cells, suggesting a direct regenerative role. 20 Upon implantation, they inhibit apoptosis of renal tubular epithelial cells and improve renal function. 5
Long-term survival of MSCs in the injured kidney has been observed in rodent models where MSCs appear to promote angiogenesis, improve renal microcirculation, and inhibit fibrotic remodeling. 5
Uterine derived MSCs
The endometrium is the lining of the uterus, and it undergoes cyclic regeneration during each estrous cycle. As a result, it is a readily accessible and renewable source of stem cells. Obtaining allogeneic uterine-derived MSCs is less invasive and less complicated than other sources, making this cell source more feasible for therapeutic applications. Uterine-derived MSCs have also been found to possess potent immunomodulatory properties including regulation of the immune response and reduction in inflammation, crucial for treating chronic inflammatory diseases.
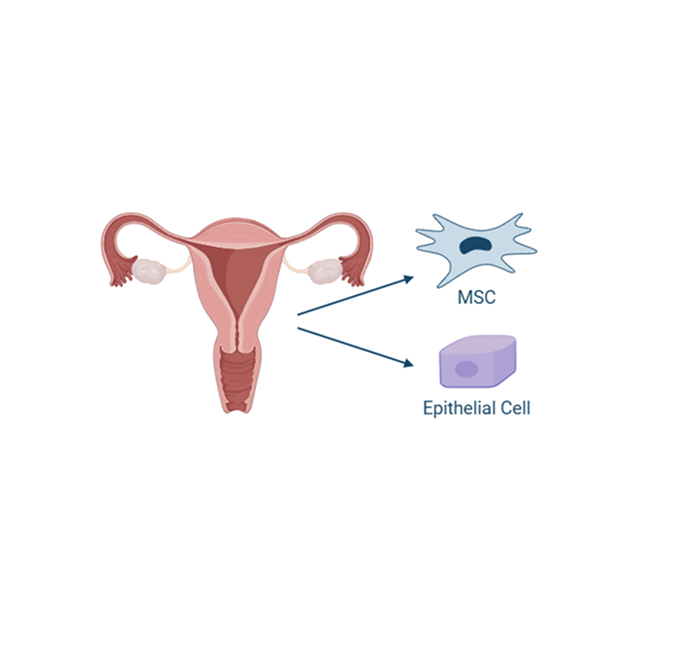
Additionally, MSCs derived from the uterus tend to have low expression of major histocompatibility complex (MHC) antigens, which makes them less likely to be recognized and attacked by the recipient’s immune system.
Available data to date indicates that UMSCs could provide a novel treatment option for cats with CKD, expanding treatment options for cats with this disease. UMSCs go beyond treatment of individual clinical signs to provide treatment of the disease at its source, potentially enhancing quality of life and preserving the caregiver/cat bond.
A deeper look into the life organ
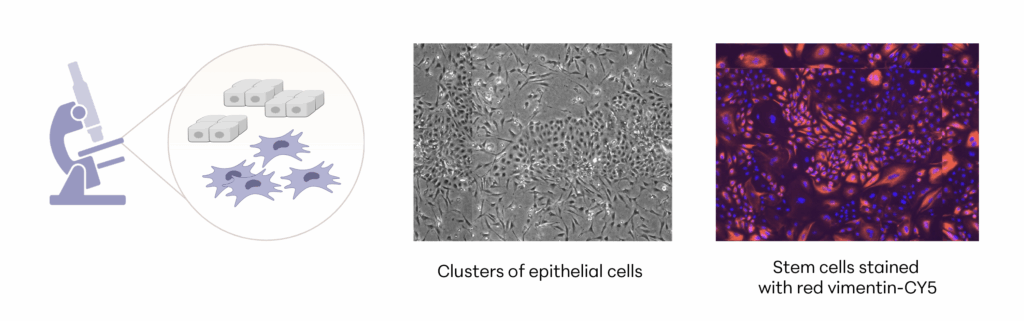
Preclinical and Clinical Insights in Feline CKD
Although most data stems from rodent models, MSC therapy is being explored in feline CKD. Early studies in cats using various administration routes (IV, intra-arterial, intrarenal) and dosages have raised minimal safety concerns, however efficacy results were mixed and follow-up durations were short (2–3 months).21–24
Allogeneic MSCs (from donor cats) are favorable over autologous sources of MSCs due to the risks related to tissue collection under anesthesia in cats who are typically older, uremic, and sometimes have other health limitations. In one controlled study, two IV injections of allogeneic uterine-derived MSCs in cats with induced CKD resulted in a ≥20% increase in GFR in 50% of subjects, along with improved water and diet intake over 6 months. 25 Larger, randomized, placebo-controlled trials are essential to establish efficacy in naturally occurring feline CKD.
Manufacturing and Potency
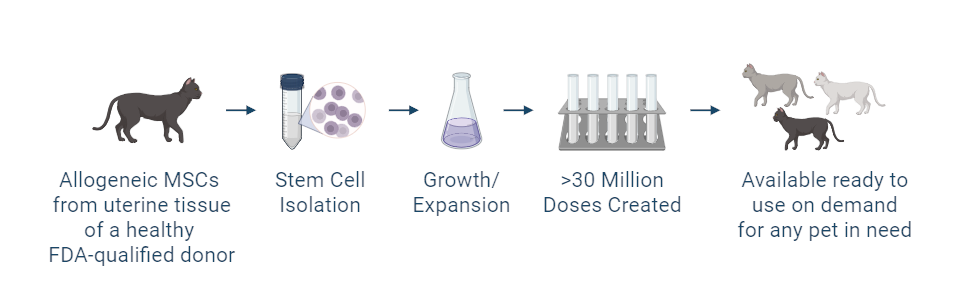
The investigational stem cells being studied are collected from an FDA-qualified feline donor during a routine spay procedure. The cells are intended to be used in other cats (same species) as an allogeneic ready to use product. The uterine-derived MSCs have been demonstrated in a matrix of assays to be potent (functional) and directed at the relevant clinical factors in feline CKD. Each batch of stem cells is manufactured according to Current Good Manufacturing Practices (CGMP) and is released with established specifications demonstrating key quality attributes for identity, purity, safety and potency of the drug product.
Allogeneic cells from a FDA qualified donor
References
- O’Neill DG, Church DB, McGreevy PD, Thomson PC, Brodbelt DC. Prevalence of disorders recorded in cats attending primary-care veterinary practices in England. Vet J. 2014;202(2):286-291. doi:10.1016/j.tvjl.2014.08.004
- Marino CL, Lascelles BDX, Vaden SL, Gruen ME, Marks SL. Prevalence and classification of chronic kidney disease in cats randomly selected from four age groups and in cats recruited for degenerative joint disease studies. J Feline Med Surg. 2014;16(6):465-472. doi:10.1177/1098612X13511446
- Quimby JM, Borjesson DL. Mesenchymal stem cell therapy in cats: Current knowledge and future potential. J Feline Med Surg. 2018;20(3):208-216. doi:10.1177/1098612X18758590
- Summers S, Quimby J. Insights into the gut-kidney axis and implications for chronic kidney disease management in cats and dogs. Vet J. 2024;306:106181. doi:10.1016/j.tvjl.2024.106181
- Chen F, Chen N, Xia C, et al. Mesenchymal Stem Cell Therapy in Kidney Diseases: Potential and Challenges. Cell Transplant. 2023;32:09636897231164251. doi:10.1177/09636897231164251
- Javard R, Grimes C, Bau‐Gaudreault L, Dunn M. Acute‐Phase Proteins and Iron Status in Cats with Chronic Kidney Disease. J Vet Intern Med. 2017;31(2):457-464. doi:10.1111/jvim.14661
- Uva A, Cavalera MA, Gusatoaia O, et al. Inflammatory Status and Chronic Kidney Disease in Cats: Old and New Inflammatory Markers—A Pilot Prospective Study. Animals. 2023;13(23):3674. doi:10.3390/ani13233674
- Syme HM, Barber PJ, Markwell PJ, Elliott J. Prevalence of systolic hypertension in cats with chronic renal failure at initial evaluation. J Am Vet Med Assoc. 2002;220(12):1799-1804. doi:10.2460/javma.2002.220.1799
- Bijsmans ES, Jepson RE, Chang YM, Syme HM, Elliott J. Changes in Systolic Blood Pressure over Time in Healthy Cats and Cats with Chronic Kidney Disease. J Vet Intern Med. 2015;29(3):855-861. doi:10.1111/jvim.12600
- Sparkes AH, Caney S, Chalhoub S, et al. ISFM Consensus Guidelines on the Diagnosis and Management of Feline Chronic Kidney Disease. J Feline Med Surg. 2016;18(3):219-239. doi:10.1177/1098612X16631234
- Chalhoub S, Langston C, Eatroff A. Anemia of Renal Disease: What it is, what to do and what’s new. J Feline Med Surg. 2011;13(9):629-640. doi:10.1016/j.jfms.2011.07.016
- White JD, Stevenson M, Malik R, Snow D, Norris JM. Urinary tract infections in cats with chronic kidney disease. J Feline Med Surg. 2013;15(6):459-465. doi:10.1177/1098612X12469522
- Polzin DJ, Bartges JW, eds. Nephrology and Urology of Small Animals. Wiley-Blackwell; 2011. doi:10.1002/9781118785546
- De Santis F, Boari A, Dondi F, Crisi PE. Drug-Dosing Adjustment in Dogs and Cats with Chronic Kidney Disease. Animals. 2022;12(3):262. doi:10.3390/ani12030262
- De Freitas MN, De Freitas MS, Da Silva TF, et al. Blood pressure and proteinuria in older cats and cats with chronic kidney disease. Vet World. Published online February 2025:527-533. doi:10.14202/vetworld.2025.527-533
- Miyamoto K. Clinical Application of Plasma Clearance of Iohexol on Feline Patients. J Feline Med Surg. 2001;3(3):143-147. doi:10.1053/jfms.2001.0125
- Elliott J, Reyes-Hughes H, Hibbert A, Blackwell E, Finch NC. Owners’ experiences of caring for cats with chronic kidney disease in the UK. J Feline Med Surg. 2025;27(4):1098612X251314769. doi:10.1177/1098612X251314769
- Yun CW, Lee SH. Potential and Therapeutic Efficacy of Cell-based Therapy Using Mesenchymal Stem Cells for Acute/chronic Kidney Disease. Int J Mol Sci. 2019;20(7):1619. doi:10.3390/ijms20071619
- Hickson LJ, Eirin A, Lerman LO. Challenges and opportunities for stem cell therapy in patients with chronic kidney disease. Kidney Int. 2016;89(4):767-778. doi:10.1016/j.kint.2015.11.023
- Dos Santos LG, Ferreira PI, Krause A. Mesenchymal stem cell transplantation: Systematic review, meta-analysis and clinical applications for acute kidney injury and chronic kidney disease in dogs and cats. Res Vet Sci. 2024;175:105313. doi:10.1016/j.rvsc.2024.105313
- Quimby JM, Webb TL, Habenicht LM, Dow SW. Safety and efficacy of intravenous infusion of allogeneic cryopreserved mesenchymal stem cells for treatment of chronic kidney disease in cats: results of three sequential pilot studies. Stem Cell Res Ther. 2013;4(2):48. doi:10.1186/scrt198
- Quimby JM, Webb TL, Gibbons DS, Dow SW. Evaluation of intrarenal mesenchymal stem cell injection for treatment of chronic kidney disease in cats: A pilot study. J Feline Med Surg. 2011;13(6):418-426. doi:10.1016/j.jfms.2011.01.005
- Quimby JM, Webb TL, Randall E, Marolf A, Valdes-Martinez A, Dow SW. Assessment of intravenous adipose-derived allogeneic mesenchymal stem cells for the treatment of feline chronic kidney disease: a randomized, placebo-controlled clinical trial in eight cats. J Feline Med Surg. 2016;18(2):165-171. doi:10.1177/1098612X15576980
- Thomson AL, Berent AC, Weisse C, Langston CE. Intra‐arterial renal infusion of autologous mesenchymal stem cells for treatment of chronic kidney disease in cats: Phase I clinical trial. J Vet Intern Med. 2019;33(3):1353-1361. doi:10.1111/jvim.15486
- Zacharias S, Welty MB, Sand TT, Black LL. Impact of allogeneic feline uterine-derived mesenchymal stromal cell intravenous treatment on renal function of nephrectomized cats with chronic kidney disease. Res Vet Sci. 2021;141:33-41. doi:10.1016/j.rvsc.2021.09.015