Mesenchymal Stromal Cell (MSC) Therapy for Feline Osteoarthritis
Gallant Pilot Trial Results
Osteoarthritis (OA) is a common and often underdiagnosed condition in cats. Recent studies suggest a probable immune-mediated component to the onset and progression of feline OA.1–3 Subtle behavioral changes including reduced activity, reluctance to jump, or changes in grooming can signal chronic joint pain that significantly impacts quality of life.4 A recent Gallant pilot clinical study evaluated the safety and efficacy of intravenous uterine-derived mesenchymal stromal cells (UMSCs) in cats with naturally occurring osteoarthritis.
Study Design
This randomized, masked, placebo-controlled clinical field study was conducted across 6 veterinary clinics in the United States and enrolled 35 client-owned cats with radiographically confirmed osteoarthritis in at least 2 joints, with pain in at least one arthritic joint. Cats were randomly assigned to receive 2 intravenous injections, administered 14 days apart, of either low-dose UMSC therapy (5 million cells), high-dose UMSC therapy (20 million cells), or saline placebo.
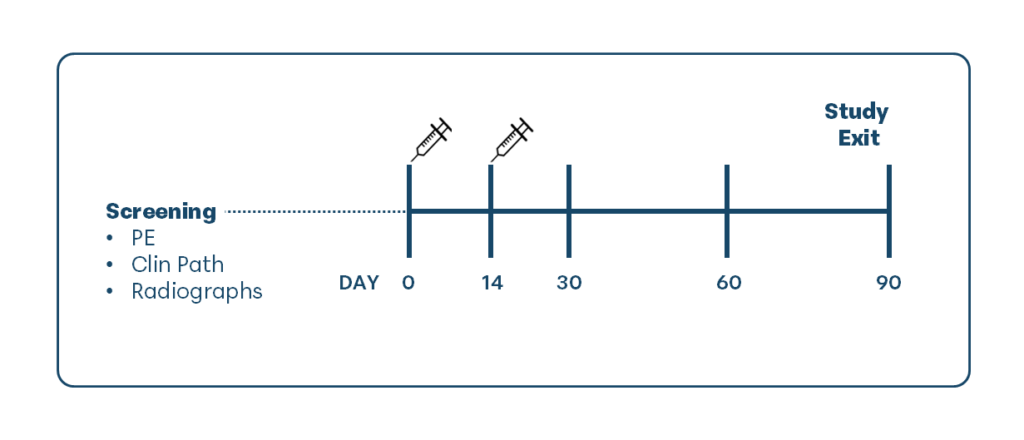
Cats were evaluated over a 90-day study period using both owner-reported and veterinarian-assessed outcomes. Owner assessments included the Client-Specific Outcome Measures (CSOM; a validated quality-of-life tool for feline osteoarthritis) and Owner Overall Assessment (OOA) of improvement. Veterinary evaluations included a Veterinary Pain Assessment (VPA) based on joint palpation and a Veterinarian Overall Assessment (VOA) of clinical response.
- CSOM: caregiver’s determination of three specific, unique activities for their cat that had become impaired since the onset of OA, evaluating each on a 5-point scale (0 = no problem, 1 = mildly problematic, 2 = moderately problematic, 3 = severely problematic, 4 = impossible) for a maximum possible total score of 12. Treatment success was defined as a reduction of at least two points in total CSOM score compared to baseline, with no increase in the score of any individual activity.
- OOA: caregiver evaluated their cat’s overall response to treatment using a categorical scale (greatly improved, mildly improved, no change, or worsened). Treatment success was defined by a response of “mildly improved” or “greatly improved.”
- VPA: veterinarian palpated at least two pre-identified affected appendicular joints or spinal segments and graded pain on a 1 to 5 scale. Treatment success was defined as a one point or greater reduction in pain score in at least one joint or spinal segment without an increase in pain score in any other joint or spinal segment.
- VOA: veterinarian assessment using a four-point categorical scale: excellent (clinical signs of OA eliminated or reduced to an inconsequential level), good (clinical signs substantially [at least 50%] reduced), fair (clinical signs minimally [less than 50%] reduced), or poor (clinical signs unaffected by therapy). Treatment success was defined as a score of “good” or “excellent,” whereas “fair” or “poor” scores were considered treatment failures.
Efficacy
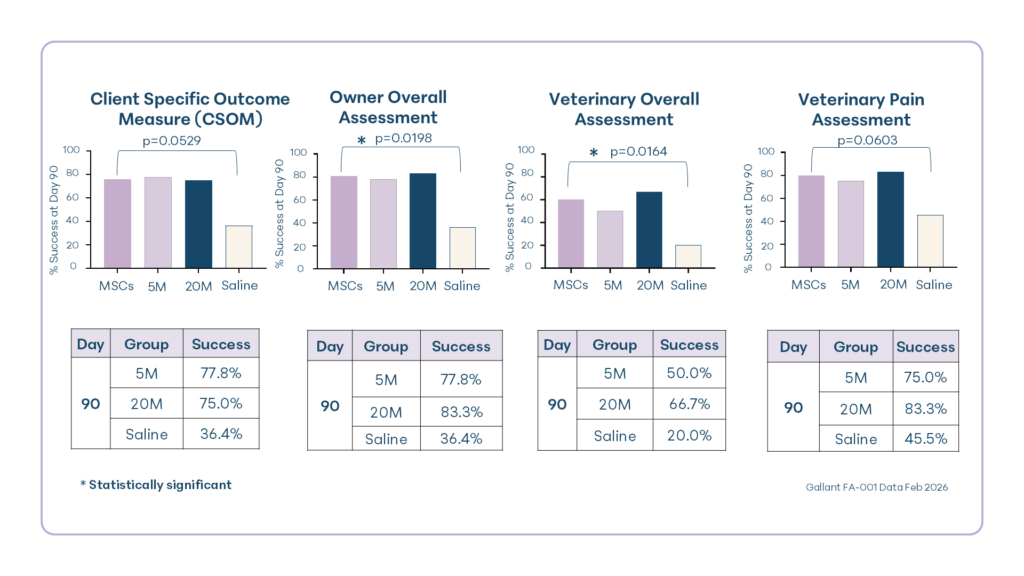
The study demonstrated statistically significant and clinically relevant improvement in both owner and veterinary outcomes in cats treated with intravenous UMSC therapy compared with placebo. Efficacy results for the 5 million and 20 million doses were similar.
By day 90, 76.2% of UMSC-treated cats showed improvement in CSOM scores, indicating better ability to perform daily activities previously impacted by osteoarthritis compared to 36.4% of placebo-treated cats. Owner overall assessments showed similar results, with 81% of treated cats reported as improved compared with 36.4% of cats receiving placebo.
Veterinary pain scores improved in 81% of treated cats, compared with 45.5% in the placebo group. Additionally, veterinarians rated 60% of treated cats as having a positive overall treatment response, compared with 20% of placebo-treated cats.
Together, these results provide evidence that intravenous (i.e. systemic) MSC therapy improves quality of life and may reduce pain in cats with osteoarthritis for at least three months following treatment.
Safety
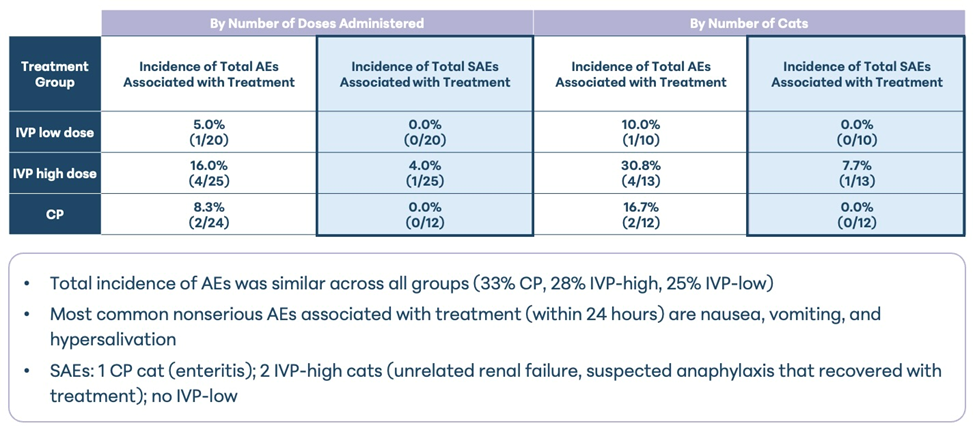
Overall, the findings indicate that intravenous administration of UMSCs is generally well tolerated in cats with naturally occurring osteoarthritis.
A summary of the AEs reported in this study are provided in the table below.
Conclusions
Results from this pilot study support efficacy at both 5 million and 20 million UMSCs. Intravenous UMSCs may provide a promising additional or alternative treatment for feline OA, a chronic debilitating disease with limited safe long term treatment options.5-7 Whereas other medications target symptoms by blocking inflammatory and pain pathways, the immunomodulatory properties of MSCs allow them to target the root cause of disease, potentially offering a disease modifying therapy in addition to control of clinical signs associated with OA.
References
- Gruen ME, Messenger KM, Thomson AE, et al. Evaluation of serum cytokines in cats with and without degenerative joint disease and associated pain. Vet Immunol Immunopathol. 2017;183:49-59. doi:10.1016/j.vetimm.2016.12.007
- Lascelles BDX, Ponnala R, Kamerling SG, Williams T. Proteomic profiling of serum in cats with naturally occurring degenerative joint disease and co-morbid conditions. Front Pain Res. 2025;6:1501932. doi:10.3389/fpain.2025.1501932
- Gao X, Lee J, Malladi S, Melendez L, Lascelles BDX, Al-Murrani S. Feline degenerative joint disease: a genomic and proteomic approach. J Feline Med Surg. 2013;15(6):6. doi:10.1177/1098612X12470652
- Lefort-Holguin M, Delsart A, Frézier M, et al. Osteoarthritis in cats: what we know, and mostly, what we don’t know. . . yet. J Feline Med Surg. 2025;27(7):1098612X251347999. doi:10.1177/1098612X251347999
- Lai X, Lin L, Chen Y, Wu L, Huang Y, Chen M. Safety assessment of frunevetmab for osteoarthritis pain in cats: disproportionality analysis of the Food and Drug Administration Animal Drug Adverse Events database. J Vet Intern Med. 2026;40(1):aalag003. doi:10.1093/jvimsj/aalag003
- FDA Center for Veterinary Medicine. Information About the Boxed Warning on Meloxicam Labels Regarding Safety Risks in Cats. U.S. Food and Drug Administration. 2024. Accessed March 10, 2026. https://www.fda.gov/animal-veterinary/product-safety-information/information-about-boxed-warning-meloxicam-labels-regarding-safety-risks-cats
- FDA Center for Veterinary Medicine. Adverse Drug Event (ADE) Database FOIA Response for Solensia (Frunevetmab). U.S. Food and Drug Administration; 2024.


