What are the Differences Between Mesenchymal Stem/Stromal Cells (MSCs) Sourced from the Patient versus MSCs Sourced from a Healthy Donor?
Veterinarians and pet parents often have many questions about using autologous (derived from the patient) mesenchymal stromal/stem cells (MSCs) versus allogeneic (derived from healthy donor) MSCs . This article provides information describing similarities and differences of autologous and allogeneic MSC therapies with regard to: safety, efficacy, consistency, ease of use, and risk for immune reaction.
Safety
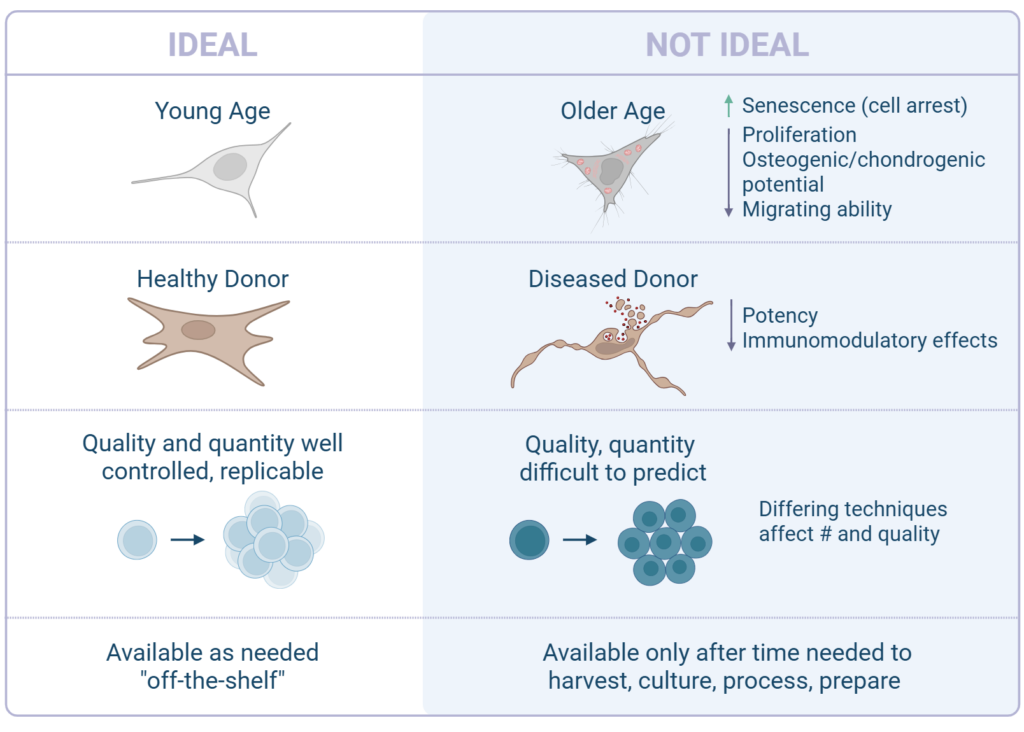
Safety profiles of autologous and allogeneic MSC therapies are generally very good. Autologous MSCs are often perceived as inherently safe because the cells originate from the patient, eliminating concerns about donor mismatch. However, autologous cells are typically harvested from older animals with active disease, and studies have shown that age and chronic inflammation can negatively affect cell health, growth, and function.1,2
Allogeneic MSCs have demonstrated comparable safety in countless clinical studies in humans and animals for a variety of conditions and in some cases, allogeneic MSCs have fewer adverse events compared with autologous cells.2–10 Adverse events across species appear to be infrequent, mild, and self-limiting and repeat dosing does not appear to be associated with immune rejection.
Efficacy
Current evidence suggests that both autologous and allogeneic MSCs can provide meaningful clinical benefit, but outcomes vary depending on cell quality, dose, and disease stage rather than cell source alone. Studies show similar improvements in tissue healing and functional outcomes with autologous and allogeneic MSCs. MSCs exert most of their therapeutic effects by releasing anti-inflammatory and regenerative signaling molecules rather than permanently engrafting in tissue. Donor health and cell vitality appear to play a larger role in treatment success than whether the cells are autologous or allogeneic, and allogeneic MSCs have shown equal or improved functional outcomes compared with autologous cells.2,3
Consistency
Consistency is a major practical consideration for MSC therapies. Autologous MSC therapies are inherently variable because cell quantity and quality depend on the individual animal’s age, disease state, and overall health. Older animals or those with chronic inflammatory conditions may yield MSCs with reduced growth, regenerative capacity, and anti-inflammatory activity.2
Allogeneic MSCs allow for careful donor selection, typically using young, healthy animals. Large-scale manufacturing under controlled conditions results in reduced batch-to-batch variability and enhanced reproducibility.1,3 This enables production of a drug product with more predictable behavior, potency, and a consistent number of cells in every dose. From a clinical standpoint, this consistency can translate into more reliable outcomes and easier treatment planning.3,11
Ease of Use
Ease of use strongly favors allogeneic MSC therapies in most clinical settings. Autologous approaches require tissue collection, followed by processing and cell expansion, which can take several weeks. This delay may be problematic for patients with acute injuries or rapidly progressing disease, and increases overall cost and logistical complexity.2 Autologous therapies also place the burden of tissue collection on the veterinarian, patient, and pet parent.
Allogeneic MSCs, by contrast, function as an off-the-shelf therapy. Cells can be prepared in advance, stored, and administered when needed. This approach reduces treatment delays, eliminates the need for invasive tissue harvest, improves consistency and accessibility across clinical environments, and dramatically lowers cost.3,8,10
Risk for Immune Reaction
Autologous MSCs are often assumed to carry no risk of immune reaction; however, changes that occur during cell culture may still trigger mild immune responses.
Allogeneic MSCs carry a theoretical risk of immune recognition, but extensive research shows that MSCs are naturally immune-modulating and express low levels of immune recognition markers (low levels of MHC class I and minimal to no MHC class II). As a result, immune reactions are uncommon and rarely clinically significant. Both veterinary and human studies report low rates of antibody formation and no clear impact on safety or effectiveness in the event of antibody formation.3,6–8,12 Advances in cell processing and immune-evasion strategies are expected to further reduce this risk and support repeat dosing when clinically indicated.1,13
1. Deuse T, et al. Progress and challenges in developing allogeneic cell therapies. Cell Stem Cell. 2025;32(4):513-528. doi:10.1016/j.stem.2025.03.004
2. Mamidi MK, et al. Allogeneic and autologous mode of stem cell transplantation in regenerative medicine: Which way to go? Med Hypotheses. 2014;83(6):787-791. doi:10.1016/j.mehy.2014.10.010
3. Li C, et al. Allogeneic vs. autologous mesenchymal stem/stromal cells in their medication practice. Cell Biosci. 2021;11(1):187. doi:10.1186/s13578-021-00698-y
4. Williams VS, et al. Clinical field study evaluating the safety and efficacy of allogeneic uterine-derived mesenchymal stem cells for refractory feline chronic gingivostomatitis. J Feline Med Surg. 2025;27(11):1098612X251385852. doi:10.1177/1098612X251385852
5. Soltero-Rivera M, et al. Mesenchymal stromal cell therapy for feline chronic gingivostomatitis: Long term experience. Front Vet Sci. 2023;10:1171922. doi:10.3389/fvets.2023.1171922
6. Colbath AC, et al. Review of cellular therapies provides new insights into the potential treatment of diverse neurologic diseases in horses and dogs. J Am Vet Med Assoc. 2024;262(S1):S121-S130. doi:10.2460/javma.23.12.0709
7. Brondeel C, et al. Review: Mesenchymal Stem Cell Therapy in Canine Osteoarthritis Research: “Experientia Docet” (Experience Will Teach Us). Front Vet Sci. 2021;8:668881. doi:10.3389/fvets.2021.668881
8. Hare JM, et al. Randomized Comparison of Allogeneic Versus Autologous Mesenchymal Stem Cells for Nonischemic Dilated Cardiomyopathy. J Am Coll Cardiol. 2017;69(5):526-537. doi:10.1016/j.jacc.2016.11.009
9. Jansen Of Lorkeers SJ, et al. Similar Effect of Autologous and Allogeneic Cell Therapy for Ischemic Heart Disease: Systematic Review and Meta-Analysis of Large Animal Studies. Circ Res. 2015;116(1):80-86. doi:10.1161/CIRCRESAHA.116.304872
10. Kasagga A, et al. Autologous Versus Allogeneic Adipose-Derived Mesenchymal Stem Cell Therapy for Knee Osteoarthritis: A Systematic Review, Pairwise and Network Meta-Analysis of Randomized Controlled Trials. Cureus. Published online April 21, 2025. doi:10.7759/cureus.82713
11. Durand N, et al. Insights into the use of mesenchymal stem cells in COVID-19 mediated acute respiratory failure. Npj Regen Med. 2020;5(1):17. doi:10.1038/s41536-020-00105-z
12. Webb TL. Stem Cell Therapy and Cats. Vet Clin North Am Small Anim Pract. 2020;50(5):955-971. doi:10.1016/j.cvsm.2020.06.002
13. Ahmed OTF, et al. The inconclusive superiority debate of allogeneic versus autologous MSCs in treating patients with HFrEF: a systematic review and meta-analysis of RCTs. Stem Cell Res Ther. 2025;16(1):175. doi:10.1186/s13287-025-04209-5


